Acorai was founded in 2019 in Helsingborg, Sweden, with one clear mission: to transform the way heart failure is managed by making intracardiac pressure monitoring non-invasive, accessible, and actionable at the point of care.
Today, over 64 million people worldwide live with heart failure, yet the gold standard for assessing their hemodynamic status – right heart catheterization – is invasive, expensive, and largely limited to specialist settings.
The company is changing that. By combining a novel array of sensors with on-device machine learning, theygive clinicians the cardiac pressure insights they need, quickly and non-invasively, without putting a catheter in a patient.
Interview with Kasper Bourdette, COO & Co-Founder of Acorai.
What are the main areas of activity?
Kasper Bourdette: Our core focus is non-invasive intracardiac pressure monitoring (ICPM) for heart failure management. Concretely, this means building and clinically validating a hardware-enabled AI platform that can estimate key hemodynamic parameters, specifically pulmonary capillary wedge pressure and right atrial pressure, from the surface of the chest. We work across three parallel tracks: clinical evidence generation, regulatory strategy, and early commercialization. Without robust clinical data, there is no regulatory pathway; without regulatory clearance, there is no commercial launch. We are engaged across all three simultaneously, which is essential to move at the pace this market needs.
What’s the news about new products/services?
K.B: The biggest clinical news during the last year has been the presentations of topline results from our CAPTURE-HF study at industry conferences. The study enrolled over 1,600 patients across 20 sites in six countries, making it the largest study of its kind. The results were strong: our device achieved an AUC of 0.821 for detecting elevated left-sided pressures and 0.852 for right-sided pressures, with sensitivity above 86% and negative predictive values reaching 97%. In practice, that “rule-out” performance is exactly what clinicians need at the bedside. Building directly on that, we enrolled our first patient in the HOSP-HF study just last week – a 1,000-patient observational study across ten centers in Europe, Asia, and the US, focused on discharge readiness in decompensated heart failure. The hypothesis is straightforward: patients sent home with residual congestion come back. If we can objectively measure that congestion at discharge, we can reduce avoidable readmissions.

What are the ranges of products/services?
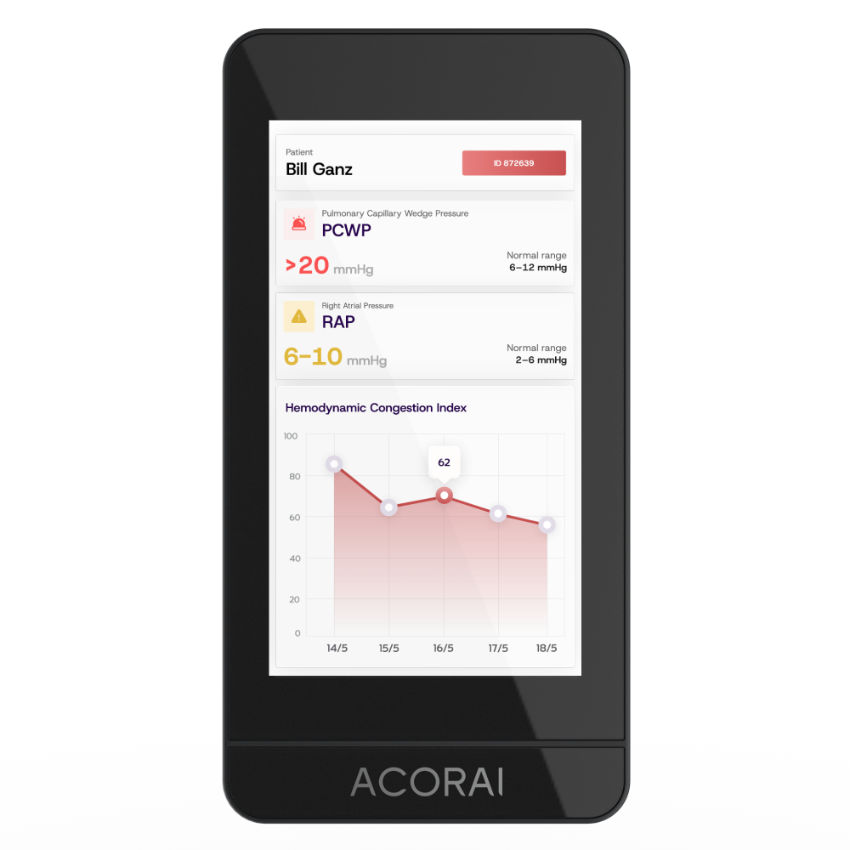
K.B: At this stage, we are focused on a single, well-defined product: the Acorai device – a compact, pocket-sized, multi-sensor platform that a nurse or generalist can use with minimal training. The user places it on the patient’s upper chest for a few minutes; the on-device machine learning models process the data and display the cardiac pressure estimates directly on screen. No wires, no invasive procedure, no specialist required. The underlying sensing architecture which we call the SAVE Sensor System integrates seismic, acoustic, visual, and electric signals. Each modality captures a different dimension of cardiac physiology, and together they feed a machine learning engine trained on one of the largest hemodynamic datasets in this clinical space. The breadth of care settings this can serve – emergency departments, outpatient clinics, general wards – is part of what makes the platform compelling.
What is the state of the market where you are currently active?
K.B: Heart failure is one of the most costly and burdensome conditions in healthcare. Hospitalization rates remain high, readmissions within 30 days are a persistent problem, and the tools available at the bedside for most clinicians are surprisingly limited. The standard of care today relies on symptoms, physical exam, and natriuretic peptides – none of which give you a direct hemodynamic picture. The invasive gold standard exists but is reserved for a small fraction of patients. There is a significant gap between what clinicians need to know and what they can actually measure. That is the market we are entering. Regulatory-wise, we received FDA Breakthrough Device Designation in 2023, which reflects the agency’s acknowledgment that this is an unmet need warranting an accelerated pathway. We are also engaged through the FDA’s TAP Pilot program, which gives us structured feedback ahead of formal submission. In Europe, we are preparing CE mark submissions in parallel.
What can you tell us about market trends?
K.B: Several trends are converging in our favor. First, there is a clear clinical and policy push toward decongestion-guided therapy – the evidence that managing heart failure patients based on actual hemodynamic data, rather than symptoms alone, leads to better outcomes is growing. Second, healthcare systems are under increasing pressure to reduce avoidable hospitalizations. Heart failure readmissions are expensive and largely preventable with better monitoring. Third, AI-powered diagnostics at the point of care are gaining acceptance and regulatory clarity. The FDA’s frameworks around AI/ML-based devices have matured considerably in recent years, which benefits companies like us who have built rigorous, prospectively validated models. And fourth, there is a broader movement toward decentralizing care – putting tools in the hands of generalists and nurses that were previously confined to catheterization labs. All of these trends point toward greater demand for exactly what we are building.
What are the most innovative products/services marketed?
K.B: The Acorai device itself is the innovation. What makes it genuinely novel is not any single sensor – it is the integration. Combining seismic, acoustic, visual, and electric signals into a coherent, real-time hemodynamic estimate is technically hard, and it required building one of the largest clinical datasets in the space to train models robust enough for clinical use. No other non-invasive device today can estimate both left- and right-sided filling pressures simultaneously, at the bedside, without any invasive access. We have filed seven patents covering various aspects of the technology. The FDA’s Breakthrough Device Designation is partly an acknowledgment of that novelty – they do not grant that designation to incremental improvements.
What estimations do you have for the second half of 2026?
K.B: The second half of 2026 will be a defining period for us. The HOSP-HF study will run actively across ten centers through 2026, and we will be accumulating the clinical utility and health economic data that regulators and payers need to see beyond technical validity. Our FDA submission is planned for 2026, and we are targeting CE mark in parallel. The second half of 2026 is when we expect to transition from a clinical-stage company to one with a clear regulatory path confirmed and early commercial infrastructure in place. That is a meaningful shift, and we are building toward it deliberately.