Rubber degradation and decomposition caused by unavoidable exposure to ozone (O3) ultraviolet light (UV) and oxygen (O2) is a prime cause of premature and excessive wear damage and a major reduction in the performance and functional lifetime of rubber conveyor belts. If the rubber compound does not contain sufficient protective chemicals, the decline effectively begins as soon as the rubber is vulcanised during the production process and not, as some may believe, something that begins to happen years down the line. The short-term and long-term effects are something that all conveyor operators need to understand but usually do not.
Although the damage is easily preventable, recent surveys show that some 80% of rubber conveyor belts sold in Europe, Africa and the Middle East have no inbuilt protection. In this special feature, technical author, Bob Nelson, explains the science and why so many manufacturers are producing conveyor belts with a severely limited lifespan.
A brief history of the science
In the late 19th and early 20th centuries, chemists determined that the cause of rubber degradation and premature failure was due to reaction with oxygen in the atmosphere referred to as ‘oxidation’. Initially, protective coatings were applied to the rubber, but these would soon wear off, and the unprotected rubber would fail.
Antioxidant chemicals such as phenols and hydroxylamine derivatives were then incorporated into rubber compound during mixing which would then bleed toward the surface over time, offering a continuous supply of protection. Chemists then discovered that rubber degradation was not purely the result of oxygen, but more as a result of exposure to ozone (O3) and ultraviolet light (UV) that attack rubber even more aggressively.
Ozone damage – causes and effects
Ground-level ozone (O3), also known as tropospheric ozone, is a trace gas in the troposphere (the lowest level of the Earth’s atmosphere) created by the photolysis of nitrogen dioxide (NO2) from sources such as automobile exhaust and industrial discharges. The scientific name is ozonolysis, which is the reaction that occurs between the molecular structure (double bonds) and ozone. The immediate result is formation of an ozonide, which then decomposes rapidly so that the double bond molecule is split.
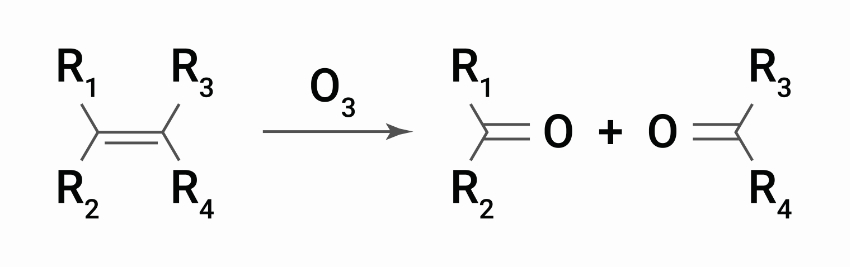
The critical step in the breakdown of molecular chains is when rubber polymers are attacked. By splitting the chain, the molecular weight drops rapidly. There comes a point when very little strength remains in the rubber. The first sign of a problem is when cracks start to appear in the surface of the rubber, sometimes referred to as ‘dry rotting’. Further attacks occur inside the newly exposed cracks, which grow steadily until they complete a ‘circuit’ and the rubber fails.

Exposure is unavoidable because even tiny traces of ozone in the air will attack the molecular structure of rubber. It increases the acidity of carbon black surfaces with polybutadiene, styrene-butadiene rubber, nitrile rubber and natural rubber being the most sensitive to degradation.
Ozone cracks form in rubber that is under tension. It is important to bear in mind that the amount of tension (critical strain) needed is extremely small. Even a belt that has not yet been fitted has a certain amount of intrinsic tension. The cracks are always oriented at right angles to the strain axis. The dynamic stress that a conveyor belt undergoes while in operation is considerable. Ozone attack occurs at the points where the strain is greatest.
Ultraviolet damage
Ultraviolet light from sunlight and artificial (fluorescent) lighting also has a seriously detrimental effect on rubber. Ultraviolet accelerates rubber deterioration because it produces photochemical reactions that promote the oxidation of the rubber surface resulting in a loss in mechanical strength. This is known as ‘UV degradation’.

What to look for
To the naked eye, rubber that has been degraded by either oxygen or ozone looks like there is a “cloudy” silver-grey film on the surface of the rubber, commonly referred to as “frosting”. This film is often mistaken for “bloom”. However, a chemical that blooms to the surface, resulting in a cloudy film, can be washed or rubbed off whereas surface degradation due to attack from either ozone, ultraviolet or oxygen cannot.

A good way to tell the difference between rubber that has been degraded by oxygen and one that has been degraded by ozone is the direction of “cracks” on the surface of the rubber. Cracks that appear due to ozone run perpendicular to the direction of the “grain” or stress applied to the rubber while cracks that appear due to oxygen are random in their orientation
Entirely preventable
Ozone, ultraviolet and oxygen damage is entirely preventable. Several years ago, Netherlands-based Fenner Dunlop were among the very first in the world to make use of new technology that enabled the effects of ozone to be tested, measured and consequently preventable. They introduced mandatory testing to EN/ISO 1431 international standards for all their rubber products. As a result, special antioxidant additives such as 2,2,4-Trimethyl-1,2-dihydroquinoline polymer (MQ), anti- degradants, antifatigue agents and antiozonants became ingredients in every rubber compound without exception.
EN/ISO 1431 testing
Resistance to ozone is measured in accordance with the EN/ISO 1431 test method in which samples of rubber are placed under tension (20% strain) inside an ozone testing cabinet and exposed to highly concentrated levels of ozone for a period of up to 96 hours (@ 40°C, 50 pphm).

Samples are examined for evidence of cracking at two-hourly intervals and the results carefully measured and recorded. Experience has determined that in order for the rubber to be regarded as adequately resistant, the pass criteria needs to be that the rubber sample does not show any signs of cracking within the 96-hour period.
Worryingly, the vast majority of samples tested typically fail within only 6 to 8 hours, which experience equates as a real time timespan of within 6 working months. Despite its crucial importance, ozone and UV resistance is very rarely, if ever, mentioned by conveyor belt manufacturers and suppliers. This is because the ingredients needed to protect the rubber and prolong its working life cost money.
Why no resistance?
Good quality antioxidants and antiozonants generally cost between €2.50/kg and €3.50/kg. A standard 400/3 4+2 abrasion belt of 200 meters and 1000 mm width typically consists of around 1700 kg of rubber compound. Total removal (non use) of antioxidants/antiozonants, anti-degradants and anti-fatigue agents generally gives a 3% or more reduction in compound cost. Although arguably a marginal saving, it is very important to those competing at the ‘grow market share at all costs’ end of the market. An additional and more sinister aspect of the non-use of protective agents is that anything that prolongs the working life of belts is not good for business so, together, they explain the widespread absence of protection.
Hidden effects
The repeated action of the mechanical stress of the conveyor belt and the frictional process from the idler triggers the oxidative chain reaction that forms a chemical process that breaks the molecular chain and activates the oxidation process This in turn magnifies a whole range of problems. Over a surprisingly short time, (literally months or even weeks without built in protection), the rubber becomes increasingly brittle and less able to stretch and contort as it should; wear resistance diminishes while transversal cracks steadily deepen and grow under the stress of passing over the pulleys and drums until catastrophic failure occurs.
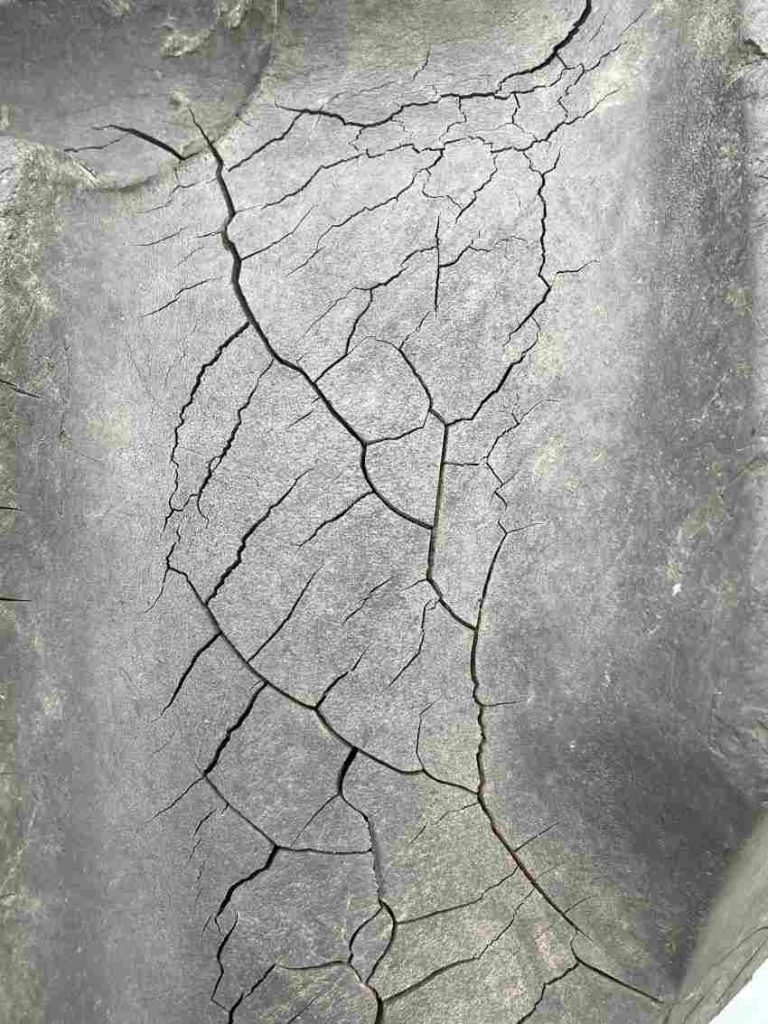
Cracks present other potential risks such as scrapers catching on them and tearing off parts of the cover. Re-splicing also becomes increasingly difficult as the adhesion properties of the rubber diminish. Environmentally, dust enters the cracks and is then shaken out on the return (underside) run causing a polluted working environment.
Conclusion
Rubber degradation and decomposition caused by exposure to ozone (O3) ultraviolet light (UV) and oxygen (O2) is far more damaging and costly both in terms of performance and working lifetime than most manufacturers would have you believe. Always insist on certification that confirms that the belt you are being offered is fully resistant in accordance with the EN ISO 1431 test method. Without this essential protection your belts will be more problematic and need to be replaced far sooner than they should be.
Author: Bob Nelson


